-
Products
- Gas analysis systems
- GAOS SENSON gas analyzers
- GAOS MS process mass spectrometry
- MaOS HiSpec ion mobility spectrometer
- MaOS AxiSpec ion mobility spectrometer
- Applications
- News
- Events
- About us
GAOS MS-20-7. TOF-MS for mud logging


Mud logging
|
Mud logging – method of qualitative and semi-quantitative gas analysis in mud during drilling or from selected samples. This method gives direct information about parameters of oil and gas reservoirs and other geological objects in well. Modern technologies of well construction, including high-speed drilling ( > 50 m/hour), horizontal drilling, geological investigation in drilling require more data from mud gas analysis:
|

|
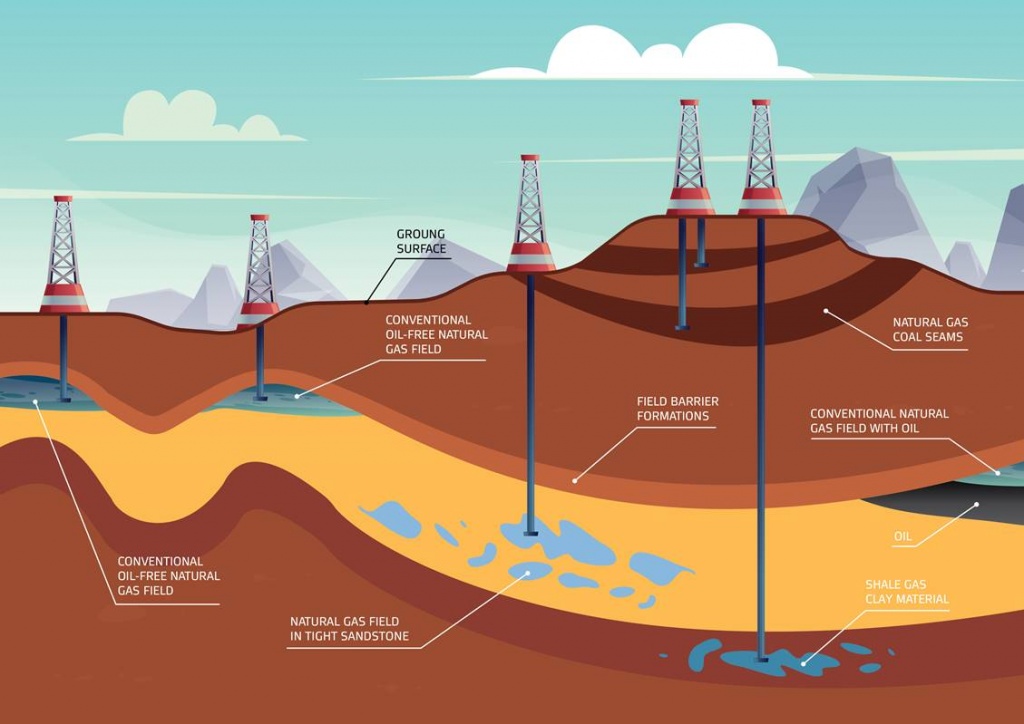
Different geology – different gas composition
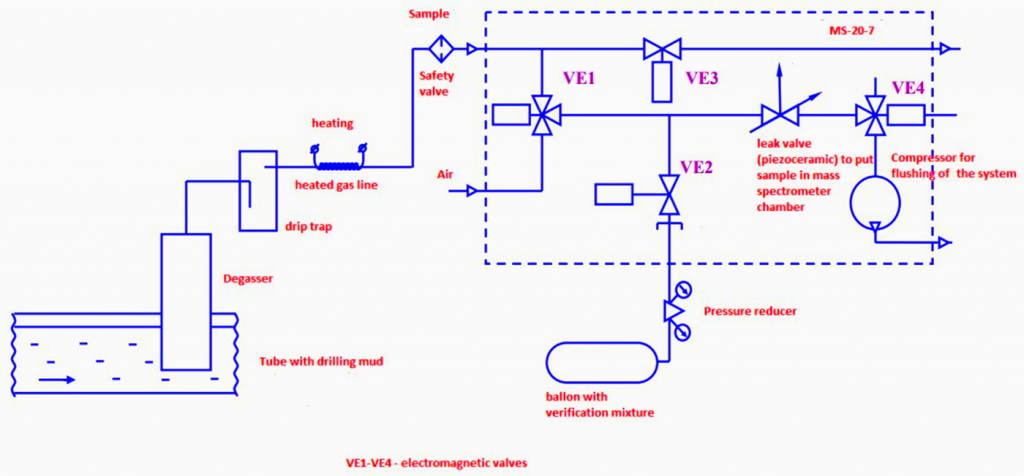
Gas scheme
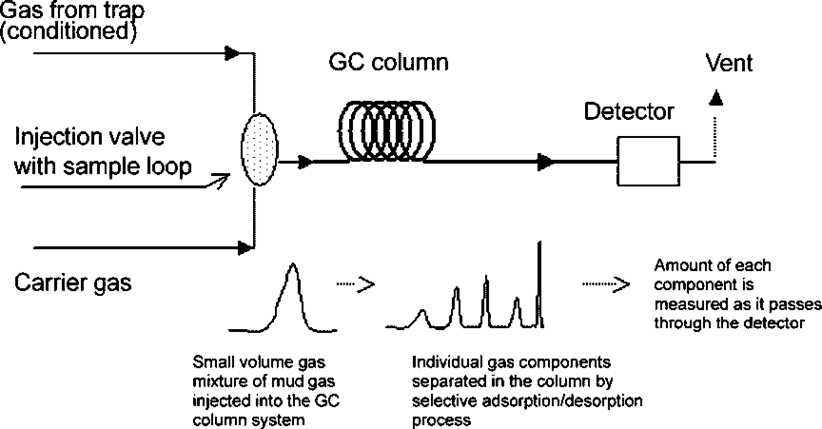
Gas Chromatography
The most widely used technique is Gas Chromatography. This instrument separates components from a mixture by selectively adsorbing and desorbing each compound at different rates. Components start to separate, depending on their affinity for the active surface of the packing or the column tubing. At the end of the column, components elute, each with unique retention time, and pass into a detector. The different detectors used to analyze the compounds eluting from the GC column include the FID, the TCD, the catalytic combustion detector (CCD), the MS detector, and the infrared (IR) absorption spectrometer.
|
Advantages |
Disadvantages |
|
|
Mass Spectrometry
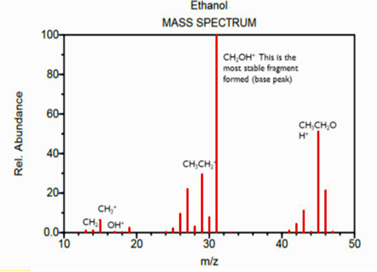
Mass spectrometry (MS) is an analytical technique that measures the mass-to-charge ratio of ions. The results are typically presented as a mass spectrum, a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is used in many different fields and is applied to pure samples as well as complex mixtures.
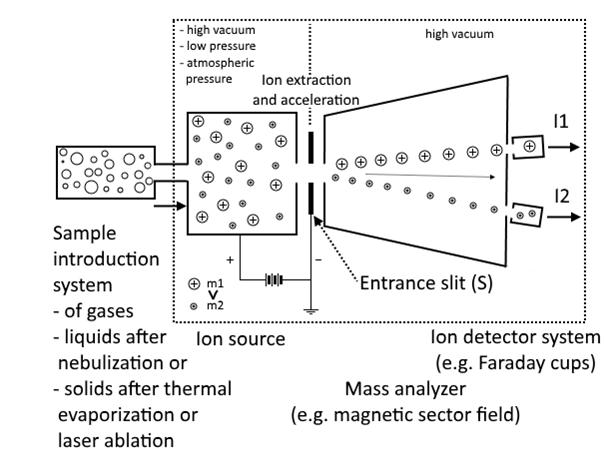
A mass spectrum is a plot of the ion signal as a function of the mass-to-charge ratio. These spectra are used to determine the elemental or isotopic signature of a sample, the masses of particles and of molecules, and to elucidate the chemical identity or structure of molecules and other chemical compounds.
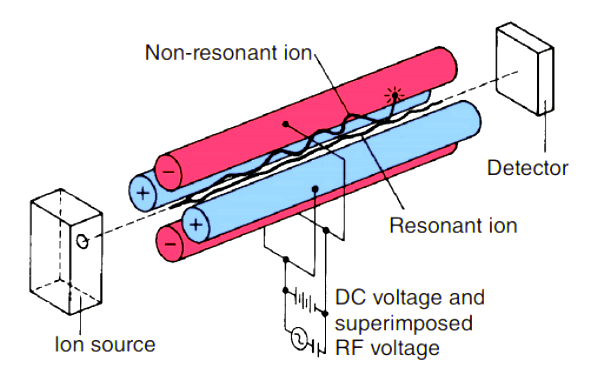
Quadrupole mass analyzers use oscillating electrical fields to selectively stabilize or destabilize the paths of ions passing through a radio frequency (RF) quadrupole field created between 4 parallel rods. Only the ions in a certain range of mass/charge ratio are passed through the system at any time, but changes to the potentials on the rods allow a wide range of m/z values to be swept rapidly, either continuously or in a succession of discrete hops.
|
Advantages |
Disadvantages |
|
|
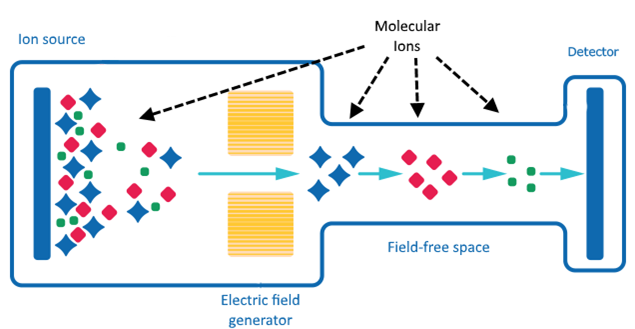
Time-of-Flight Mass Spectrometry
|
Advantages |
Disadvantages |
|
|
|
 |
 |
|
Analytical unit |
Electronic unit |
|
Analytical parameters
| Parameters | Value |
| Range of masses, Da | 1-200 |
| Resolution, M/ΔM | 200 |
|
Registration channels -Analogue -Counter |
16 16 |
| Analyzed components | CH4, C2H6, C3H8, i-C4H10, C4H10, C5H12, C6H14, H2, He, N2, Ar, O2, CO2, H2S, SO2 |
| Analysis time, no more than, s | 2 |
| Dynamic range, % vol. | 10-4-100% |
| Calibration | Automatic |
| Baseline correction | Automatic |
Design
Control software
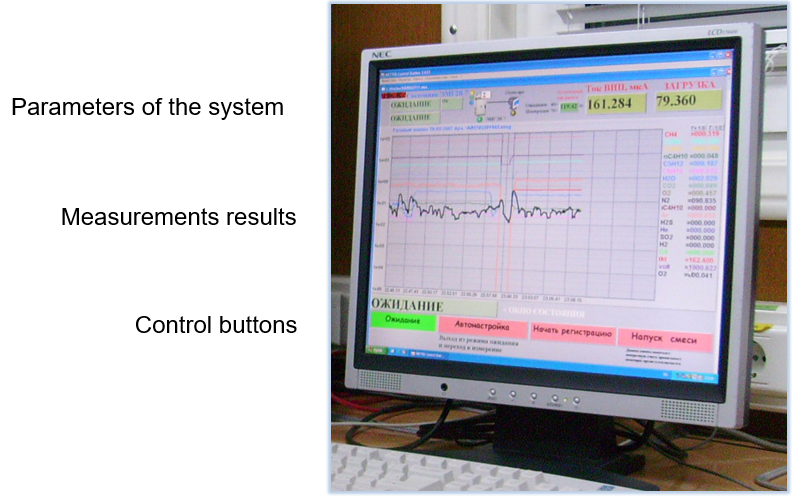
Analytical task
| Components | Concentration range, % vol. | |
| CH4 | 5,0 * 10-4 | 10,0 |
| C2H6 | 5,0 * 10-4 | 1,0 |
| C3H8 | 5,0 * 10-4 | 1,0 |
| C4H10 | 5,0 * 10-4 | 1,0 |
| C5H12 | 2,0 * 10-4 | 0,1 |
| C6H14 | 2,0 * 10-4 | 0,1 |
| N2 | 0,1 | 80,0 |
| Ar | 1,0 * 10-2 | 2,0 |
| He | 1,5 * 10-3 | 1,0 |
| O2 | 0,1 | 25,0 |
| CO2 | 0,1 | 1,0 |
| SO2 | 0,1 | 1,0 |
| H2S | 1,0*10-2 | 2,0 |
Results
|
Components |
Concentration, % vol. |
Difference, % vol. |
SD, % vol. |
|
|
CRM |
Measurement |
|||
|
СН4 |
10,31 ± 0,01 |
10,297 |
0,013 |
0,042 |
|
С2Н6 |
1,99 ± 0,09 |
2,075 |
0,085 |
0,004 |
|
С3Н8 |
0,99 ± 0,03 |
1,024 |
0,034 |
0,0012 |
|
С4Н10 |
0,533 ± 0,050 |
0,583 |
0,050 |
0,0002 |
|
i- С4Н10 |
0,054 ± 0,006 |
0,048 |
0,006 |
0,0005 |
|
С5Н12 |
0,0098 ± 0,0006 |
0,00989 |
0,00009 |
0,00004 |
|
С6Н14 |
0,000117 ± 0,00007 |
0,00118 |
0,00001 |
0,00001 |